PHILADELPHIA, U.S./CHENGDU, China: Periodontitis remains the sixth most prevalent infectious disease worldwide, and the most common cause of tooth loss despite significant advancements in oral health care. A new cell type discovered in the gingival epithelium that helps protect against harmful bacteria has renewed the interest in the immunological regulation of periodontal diseases.
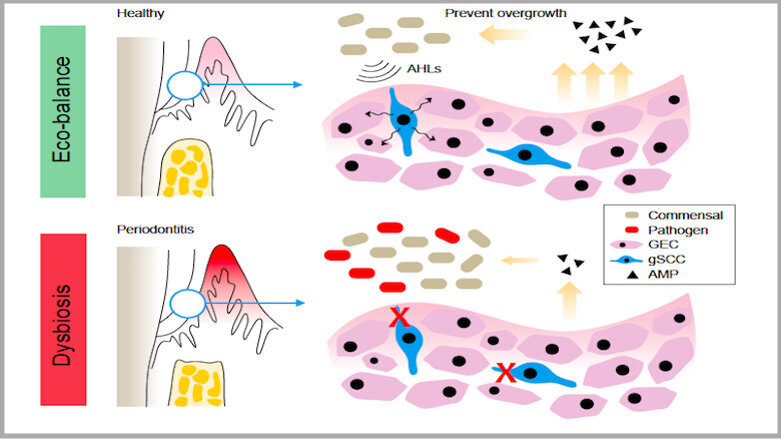
Periodontitis is a chronic inflammation of the tooth-supporting tissues induced by a bacterial infection. Recent studies have shown that periodontitis results from a condition known as polymicrobial dysbiosis, which disturbs the ecologically balanced oral microbiota needed for maintaining normal periodontal homeostasis. The host innate immune system remains highly active in order to maintain the homeostasis in healthy periodontal tissue; however, an imbalance or disruption in innate immunity also contributes to the destruction of periodontal tissue. The host-microbiota interactions that determine periodontal homeostasis are complex and remain poorly defined. We have very limited knowledge of the specific host receptors that detect pathogenic oral bacteria.
However, this study appears to have taken us closer to the point of understanding the receptors and molecules that can influence the onset of periodontitis and its management.
Researchers from Monell Chemical Senses Center, a nonprofit independent scientific institute, collaborated with the scientists from Sichuan University in Chengdu in China and examined the gingivae of mice, where they found a cell type called solitary chemosensory cells (SCCs) that expressed different types of taste receptors as well as the protein gustducin. SCCs, which have previously been found in the urinary tract, the gut and the nasal cavities, function by sensing irritants and bacteria.
The study showed that when the researchers genetically removed gustducin and/or SCCs from the mice’s gingivae, often pathogenic oral bacteria quickly grew in numbers, resulting in periodontitis. In contrast, when they stimulated the bitter taste receptors in SCCs, it promoted the production of antimicrobial molecules.
Overall, mice without gustducin in their SCCs harboured more potentially harmful oral microbiome than those with gustducin present in their SCCs. Most importantly, the researchers identified these differences in oral flora compositions much before any periodontal bone loss occurred, which implies that the presence or absence of gustducin in SCCs could be regarded as a forerunner to periodontitis and help us in the early identification of the disease.
“Our study adds to a growing list of tissues we now know contain SCCs and indicates that the common molecular pathways in gum SCCs are involved in the regulation of oral microbiota,” said Dr Marco Tizzano, a researcher & the co-author of this study at Monell Chemical Senses Center. “In the absence of taste signalling in the gums, the oral microbiome changed in mice without gustducin.”
Topical treatment with bitter-tasting denatonium could increase the expression of antimicrobial
peptides against ligature-induced periodontitis in gustducin positive mice, and not in others.
The research team, based on this study and other unpublished work relating to humans, has suggested that periodontal SCCs in humans may play a similar regulatory role in regard to our own oral microbiomes.
The study, titled “Gingival solitary chemosensory cells are immune sentinels for periodontitis,” was published online on Oct. 3, 2019, in Nature Communications.
Tags:
LOUISVILLE, Ky., US: Researchers from the US have recently examined the role that oral microbes play in regulating antiviral responses in the oral cavity. ...
This article is Part 2 in our series on dental litigation in India. In Part 1, we examined the rising trend of dental litigations and the increasing legal ...
SENDAI, Japan: Periodontitis is a common oral disease that affects the oral health of many people with serious effects. A new research from Japan proposes a...
PHILADELPHIA, US: It was previously believed that Streptococcus mutans was the main microorganism responsible for dental caries. However, in a recent study,...
World No Tobacco Day is observed on 31st of May. The theme for this year is “Get ready for plain packaging”. Armed with stricter laws and an ...
Vaccine development against SARS-CoV-2 would be greatly facilitated by the identification of immunological parameters that correlate with protection in ...
Periodontitis is a chronic oral inflammatory condition, commenced by the accumulation of pathogenic dental plaque biofilm above and below the gingival ...
One of the most critical questions the world is waiting for an answer is if the human body can mount a protective response that provides a lasting immunity ...
ChairsideCAD, available from exocad, is the choice of software for leading manufacturers of dental CAD/CAM systems, according to the company, and it has ...
Scientists have just unveiled a revolutionary breakthrough—Anthrobots— self-assembling tiny robots made from your own cells that can repair damaged ...
Live webinar
Wed. 3 June 2026
10:30 pm IST (New Delhi)
Live webinar
Thu. 4 June 2026
11:30 pm IST (New Delhi)
Live webinar
Mon. 8 June 2026
9:30 pm IST (New Delhi)
Live webinar
Mon. 8 June 2026
10:30 pm IST (New Delhi)
Dr. Anthony Mak B.D.S, Prof. Marleen Peumans
Live webinar
Mon. 8 June 2026
11:30 pm IST (New Delhi)
Live webinar
Wed. 10 June 2026
8:30 pm IST (New Delhi)
Live webinar
Wed. 10 June 2026
11:30 pm IST (New Delhi)
Nacho Fernández-Baca DDS, MSc



 Austria / Österreich
Austria / Österreich
 Bosnia and Herzegovina / Босна и Херцеговина
Bosnia and Herzegovina / Босна и Херцеговина
 Bulgaria / България
Bulgaria / България
 Croatia / Hrvatska
Croatia / Hrvatska
 Czech Republic & Slovakia / Česká republika & Slovensko
Czech Republic & Slovakia / Česká republika & Slovensko
 France / France
France / France
 Germany / Deutschland
Germany / Deutschland
 Greece / ΕΛΛΑΔΑ
Greece / ΕΛΛΑΔΑ
 Hungary / Hungary
Hungary / Hungary
 Italy / Italia
Italy / Italia
 Netherlands / Nederland
Netherlands / Nederland
 Nordic / Nordic
Nordic / Nordic
 Poland / Polska
Poland / Polska
 Portugal / Portugal
Portugal / Portugal
 Romania & Moldova / România & Moldova
Romania & Moldova / România & Moldova
 Slovenia / Slovenija
Slovenia / Slovenija
 Serbia & Montenegro / Србија и Црна Гора
Serbia & Montenegro / Србија и Црна Гора
 Spain / España
Spain / España
 Switzerland / Schweiz
Switzerland / Schweiz
 Turkey / Türkiye
Turkey / Türkiye
 UK & Ireland / UK & Ireland
UK & Ireland / UK & Ireland
 International / International
International / International
 Brazil / Brasil
Brazil / Brasil
 Canada / Canada
Canada / Canada
 Latin America / Latinoamérica
Latin America / Latinoamérica
 USA / USA
USA / USA
 China / 中国
China / 中国
 Pakistan / Pākistān
Pakistan / Pākistān
 Vietnam / Việt Nam
Vietnam / Việt Nam
 ASEAN / ASEAN
ASEAN / ASEAN
 Israel / מְדִינַת יִשְׂרָאֵל
Israel / מְדִינַת יִשְׂרָאֵל
 Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
 Middle East / Middle East
Middle East / Middle East















































To post a reply please login or register