This was a challenging case with the combined issues of a failed root canal therapy and an iatrogenic perforation of the floor of the pulp chamber. Though initially the prognosis could not be evaluated effectively simply because of non-availability of peer-reviewed literature of similar cases for citation, but the case was taken on owing to the patient’s desire to salvage the natural tooth. The importance of magnification and the ergonomic use of the dental operating microscope cannot be more clearly demonstrated than in this case.
Perforations are quite unpredictable to deal with, and the prognosis depends upon an array of criteria, such as the location, size, time lapse between the occurrence of the perforation and the repair, pre-existing infection at the site and material used for the repair. Though there are few studies available regarding the combination of mineral trioxide aggregate (MTA) and platelet-rich fibrin (PRF), owing to their biocompatible and autologous nature, respectively, and having a follow-up of the case of over one year and a few other studies with similar follow ups, the combination appears satisfactory.
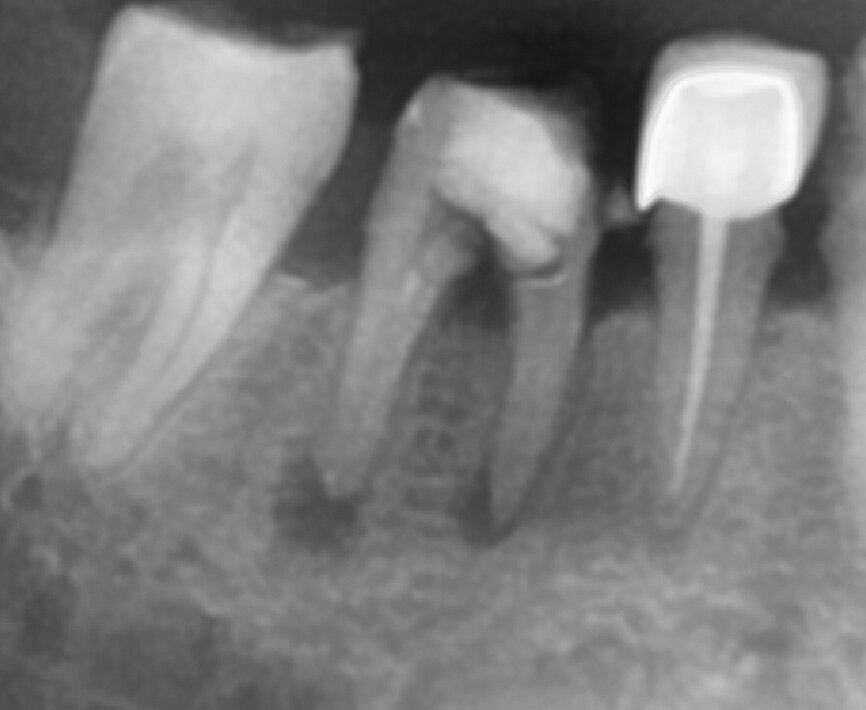
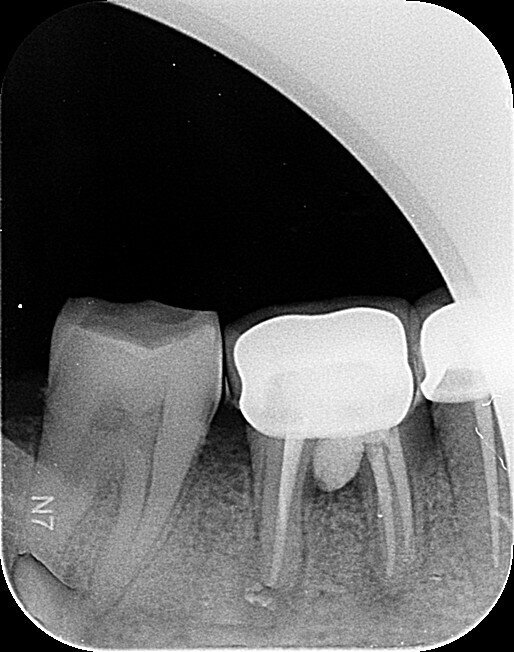
Fig.1a: Pre-op radiograph taken by the referring dentist.
Fig.1b: Pre-op radiograph taken by the author in relation to tooth #46 (January 2019).
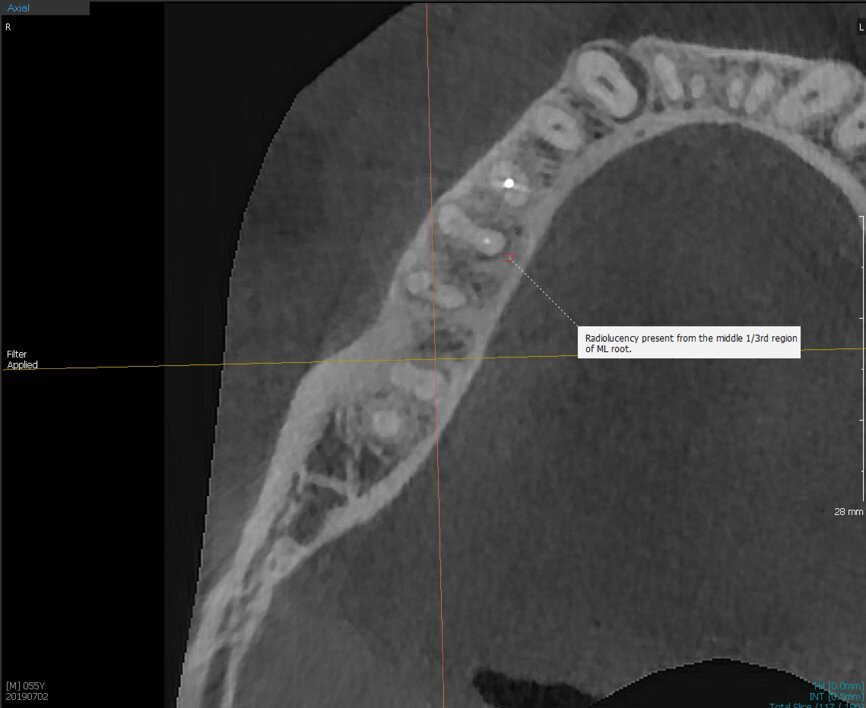
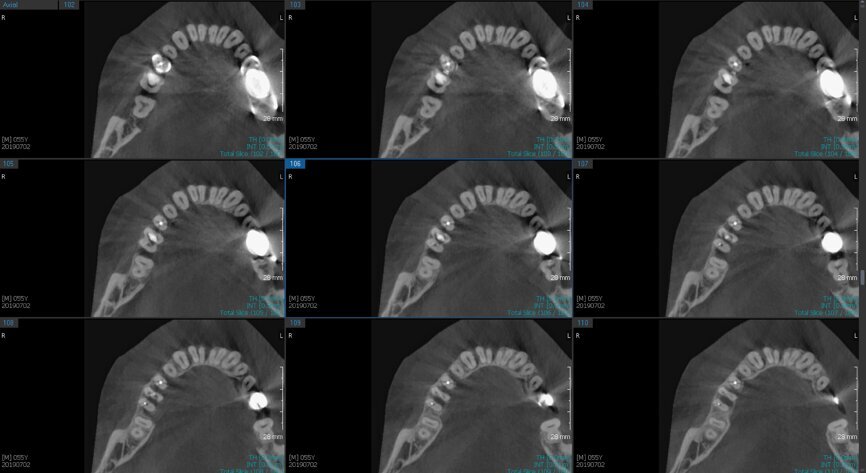
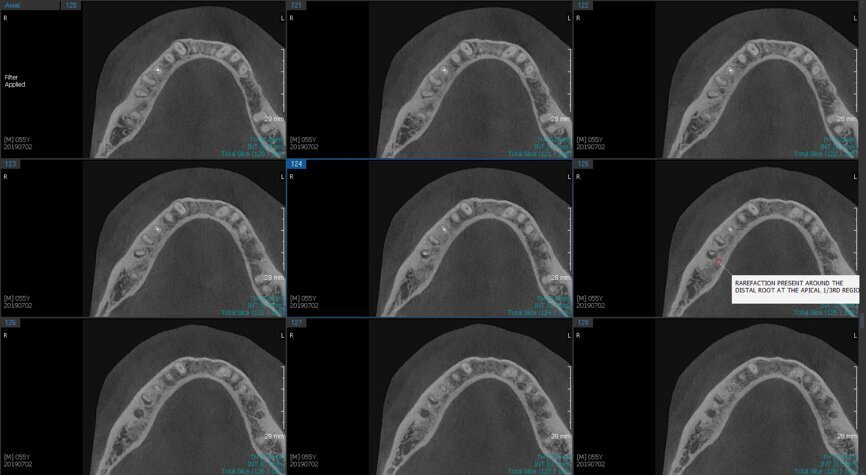
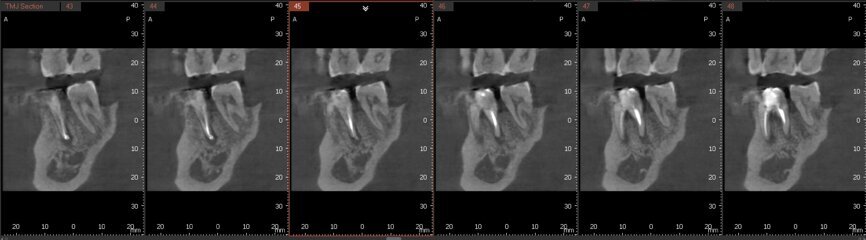
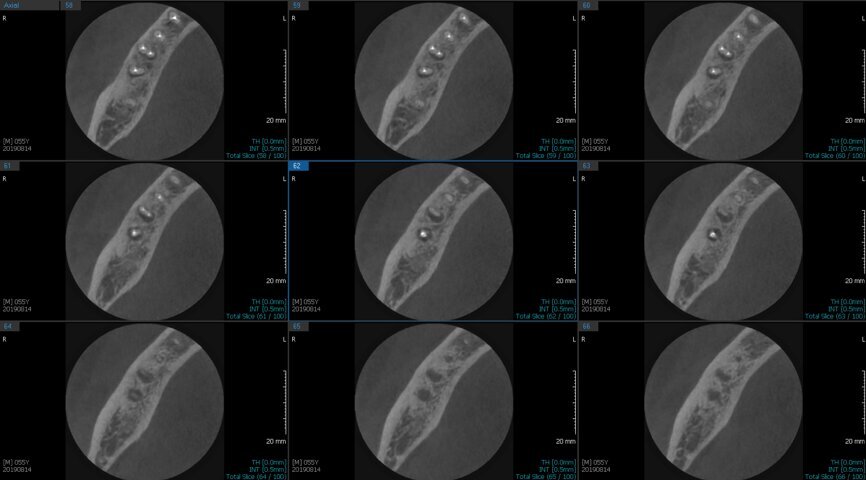
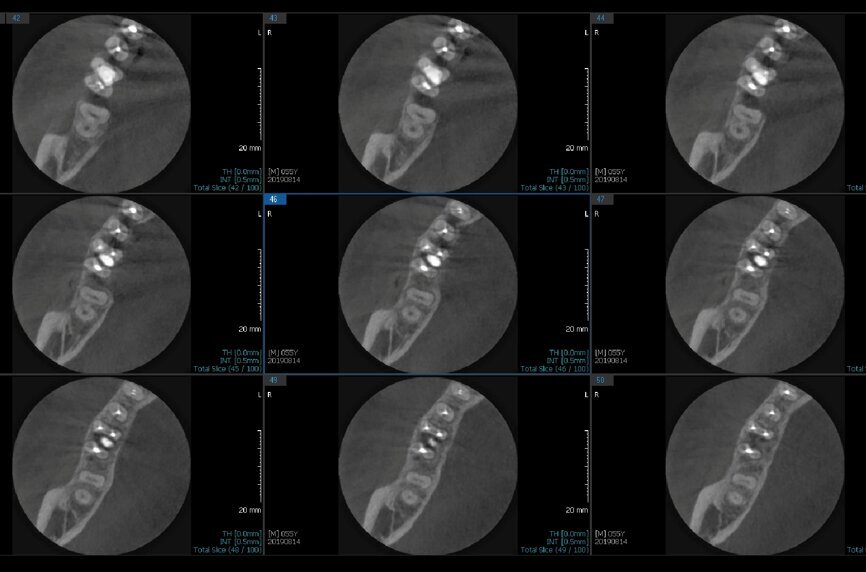
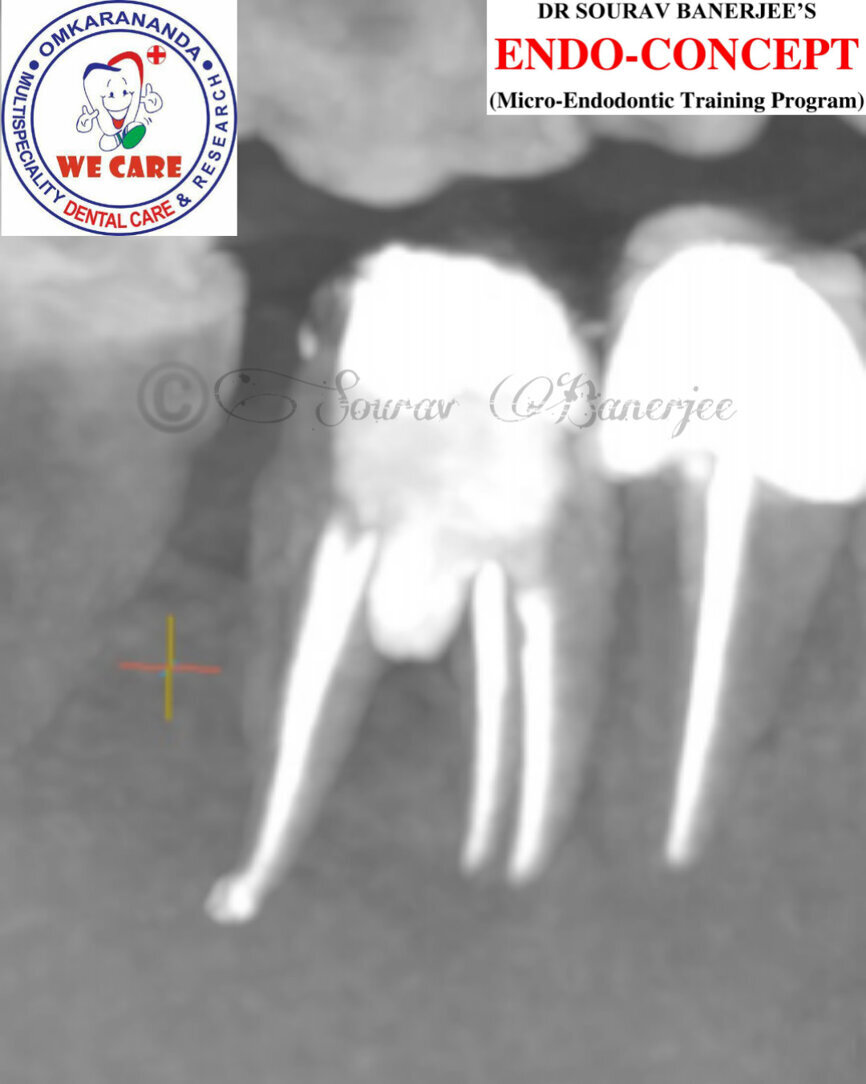
Fig.2a: Pre-op CBCT images of tooth #46: No obturation material in the distal and mesiobuccal canal (a); scanty obturation of the canals and breach of the floor of the pulp chamber, no obturation beyond a few millimetres down the orifice (b & c); radiolucency in the furcation area and periapical region of both roots (d -g).
Fig.2b: Pre-op CBCT images of tooth #46: No obturation material in the distal and mesiobuccal canal (a); scanty obturation of the canals and breach of the floor of the pulp chamber, no obturation beyond a few millimetres down the orifice (b & c); radiolucency in the furcation area and periapical region of both roots (d -g).
Fig.2c: Pre-op CBCT images of tooth #46: No obturation material in the distal and mesiobuccal canal (a); scanty obturation of the canals and breach of the floor of the pulp chamber, no obturation beyond a few millimetres down the orifice (b & c); radiolucency in the furcation area and periapical region of both roots (d -g).
Fig.2d: Pre-op CBCT images of tooth #46: No obturation material in the distal and mesiobuccal canal (a); scanty obturation of the canals and breach of the floor of the pulp chamber, no obturation beyond a few millimetres down the orifice (b & c); radiolucency in the furcation area and periapical region of both roots (d -g).
Fig.2e: Pre-op CBCT images of tooth #46: No obturation material in the distal and mesiobuccal canal (a); scanty obturation of the canals and breach of the floor of the pulp chamber, no obturation beyond a few millimetres down the orifice (b & c); radiolucency in the furcation area and periapical region of both roots (d -g).
Fig.2f: Pre-op CBCT images of tooth #46: No obturation material in the distal and mesiobuccal canal (a); scanty obturation of the canals and breach of the floor of the pulp chamber, no obturation beyond a few millimetres down the orifice (b & c); radiolucency in the furcation area and periapical region of both roots (d -g).
Fig.2g: Pre-op CBCT images of tooth #46: No obturation material in the distal and mesiobuccal canal (a); scanty obturation of the canals and breach of the floor of the pulp chamber, no obturation beyond a few millimetres down the orifice (b & c); radiolucency in the furcation area and periapical region of both roots (d -g).
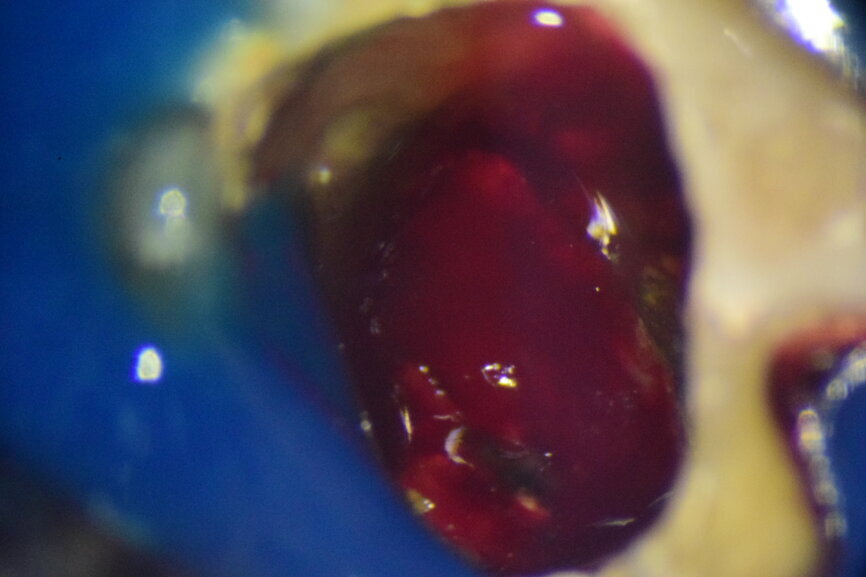
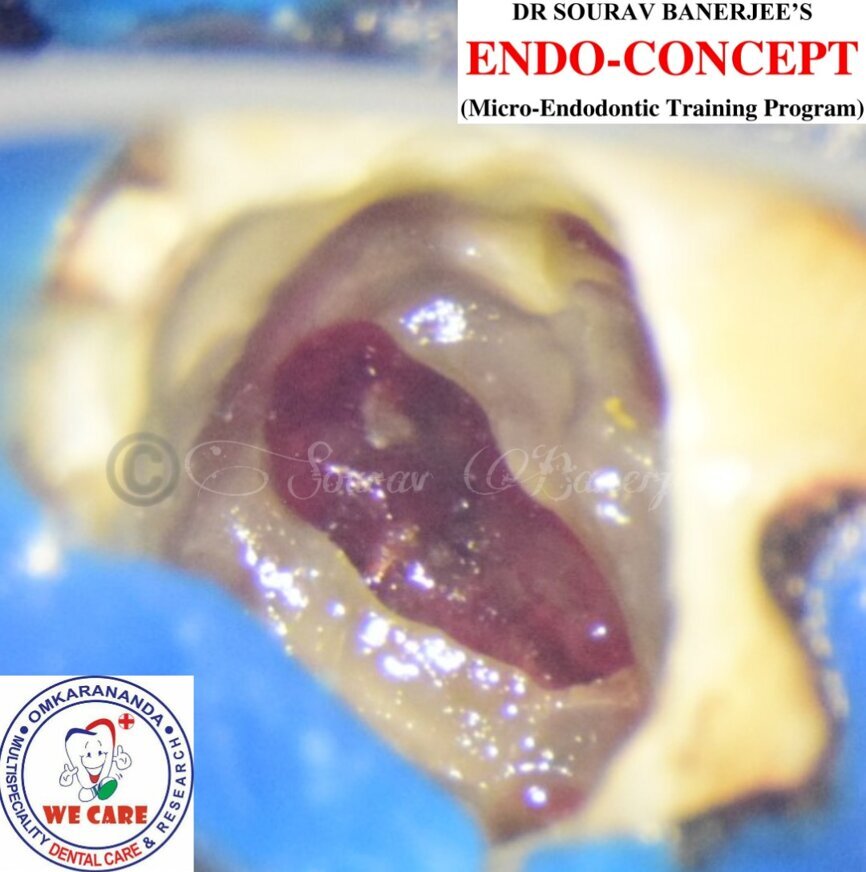
Fig. 3: Intra-op image of tooth #46 captured under the dental operating microscope at 16× magnification.
Fig. 4: Intra-op image of tooth #46 captured under the dental operating microscope at 16× magnification showing the clean perforation defect prior to its sealing with MTA and PRF.
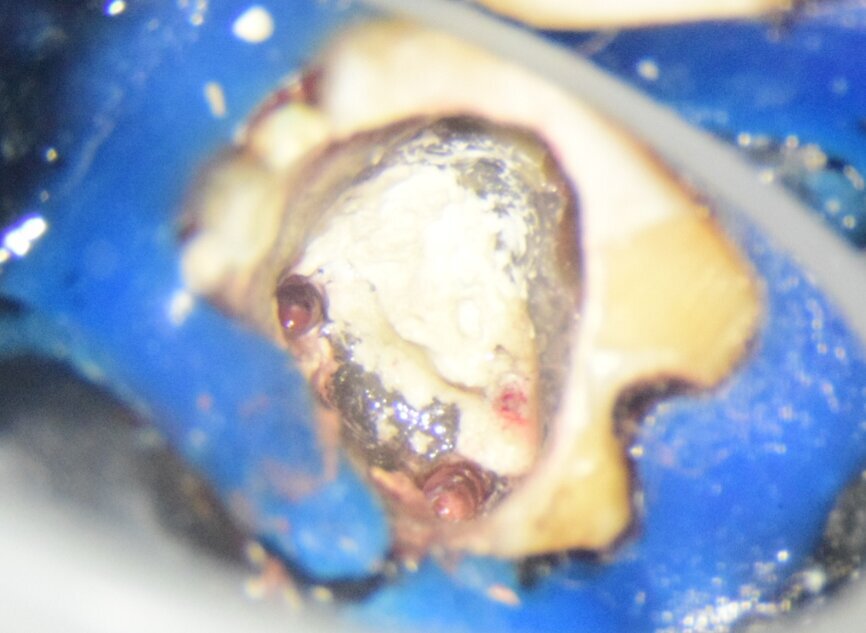
Fig. 5: Intra-op image of tooth #46 under the dental operating microscope at 16× magnification showing the sealed pulpal floor perforation with MTA and the obturated mesial canals with room for orifice plugs for the core material.
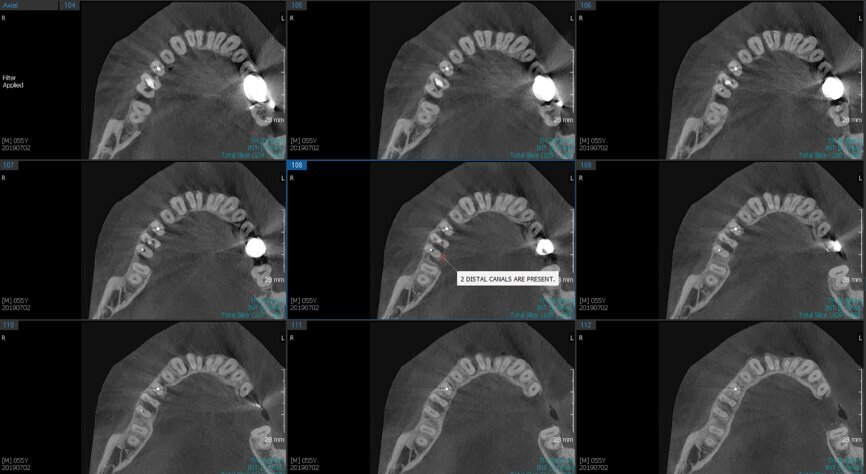
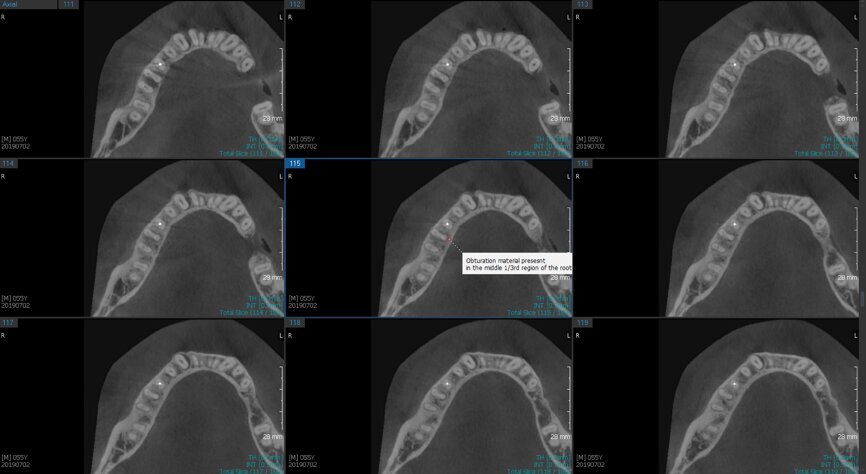
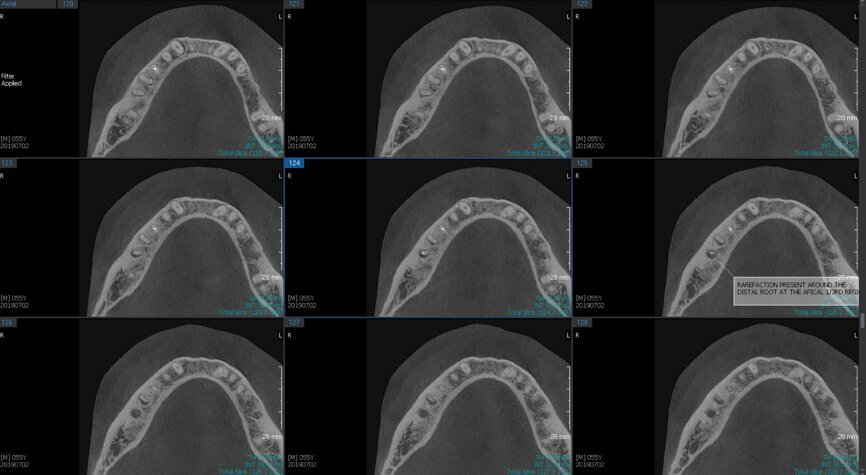
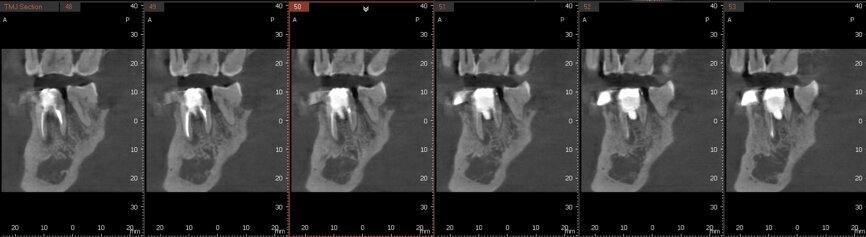
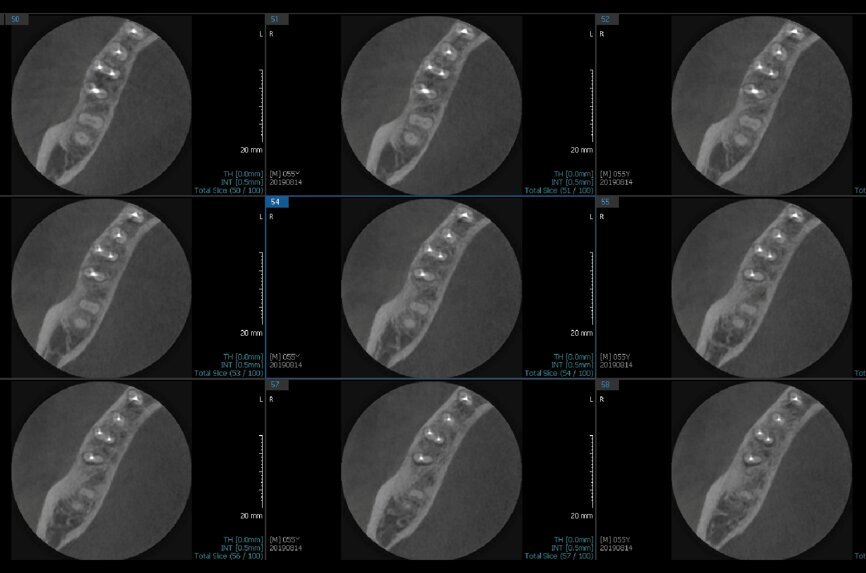
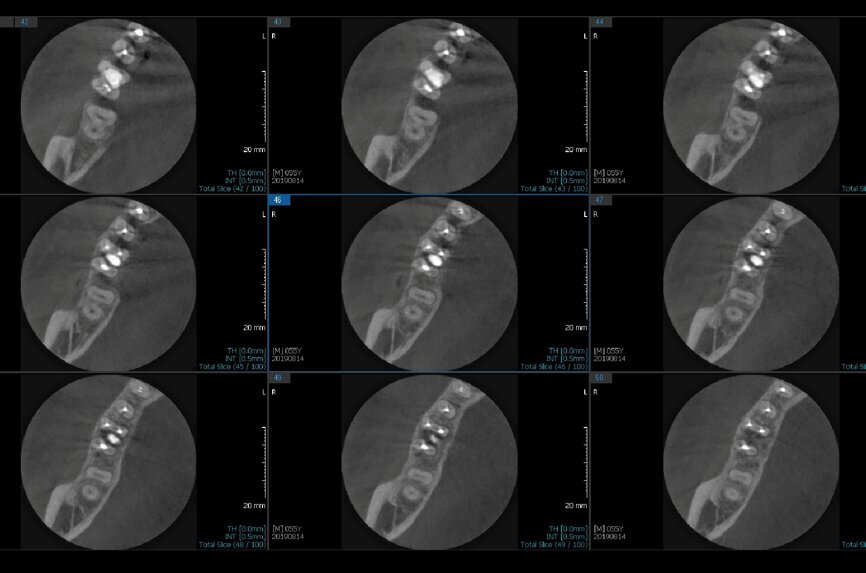
Fig. 6a: Post-op CBCT images of tooth #46 showing adequately obturated canals at all levels to the working length and sealing of the furcation defect.
Fig. 6b: Post-op CBCT images of tooth #46 showing adequately obturated canals at all levels to the working length and sealing of the furcation defect.
Fig. 6c: Post-op CBCT images of tooth #46 showing adequately obturated canals at all levels to the working length and sealing of the furcation defect.
Fig. 6d: Post-op CBCT images of tooth #46 showing adequately obturated canals at all levels to the working length and sealing of the furcation defect.
Fig. 6e: Post-op CBCT images of tooth #46 showing adequately obturated canals at all levels to the working length and sealing of the furcation defect.
Fig. 6f: Post-op CBCT images of tooth #46 showing adequately obturated canals at all levels to the working length and sealing of the furcation defect.
Fig. 6g: Post-op CBCT images of tooth #46 showing adequately obturated canals at all levels to the working length and sealing of the furcation defect.
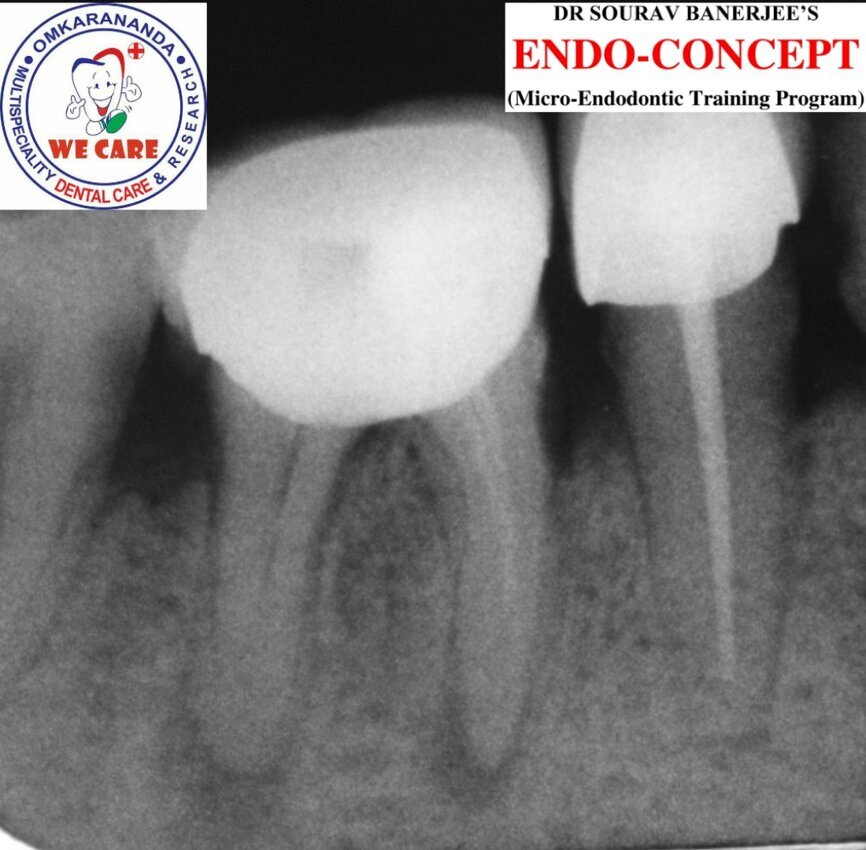
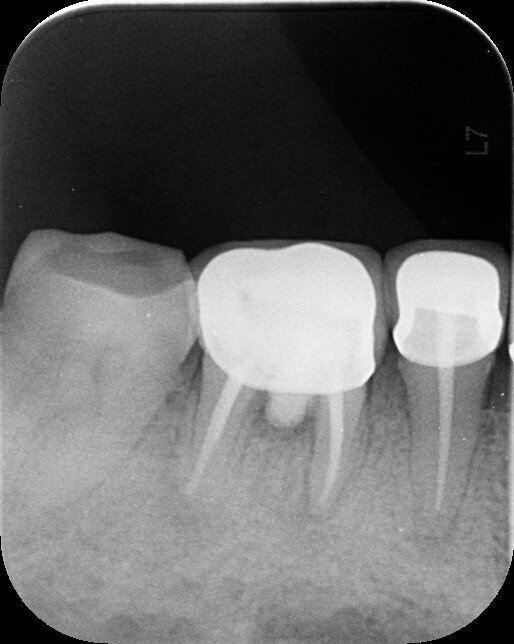
Fig.7a: Six-month follow-up (August 2019).
Fig. 7b: Fourteen-month follow-up (May 2020).
Case report
A 56-year-old male patient was referred to our practice by a general dentist. The patient had symptomatic apical periodontitis of tooth #46 and a continuous dull aching pain that radiated to the right side of his face and neck. I took a detailed history of the case and came to learn that the tooth had first been treated two years before, and when the patient developed pain in the same tooth, he had visited a second dentist, who took an intra-oral periapical radiograph and suggested a retreatment. That dentist had performed the coronal disassembly and attempted a retreatment, but failed to locate the mesiolingual canal, which the first general dentist too had been unable to locate. Subsequently, after several appointments to locate the mesiolingual canal, the second general dentist had perforated the floor of the pulp chamber. He did not inform the patient of this, but placed a temporary restoration and kept him on oral antibiotics and analgesics. After several days, the patient reported to the dentist with pain and was advised to have the tooth extracted and replaced with an implant, as it could not be saved. Since the patient was unwilling to do so, however, the dentist referred him to our practice.
When I had a close look at the preoperative radiograph from the referring dentist (Fig. 1a), I observed that there was insufficient obturation of the mesial canals and almost no obturation of the distal canal. Both roots displayed periapical radiolucency. The clinical examination revealed a Grade I mobility, and the tooth was tender to percussion. The periodontal examination ruled out any pockets and loss of attachment. We took an intra-oral periapical radiograph (Fig. 1b), from which we observed the loss of coronal structure and a large radiolucent area over the furcation area and the periapical area of both the mesial and the distal roots. A CBCT scan of tooth #46 was advised to check for any details that may have been missed in the intra-oral periapical radiograph (Figs. 2a–g). From the CBCT scan, it could be observed that there was inadequate obturation of the distal canal on the horizontal plane, no obturation beyond the middle third in the mesial canals and a breach of the floor of the pulp chamber. The questionable prognosis was explainedto the patient, and written consent was obtained.
An inferior alveolar nerve block was administered, and a dental dam was placed. When the temporary restoration was removed, blood began oozing out from the pulp chamber. After initial haemostasis, a large blood clot could be observed on the floor of the pulp chamber (Fig. 3). The clot and the underlying granulation tissue were removed with a sharp spoon excavator, and the pulp chamber was irrigated with saline. Then a sterile cotton pellet soaked in 1% sodium hypochlorite was placed in the chamber for two to three minutes until the oozing of the blood stopped.
The mesiolingual canal was located under the dental operating microscope (Sanma Medineers) using an RS-1 ultrasonic tip (KaVo Kerr) at the #5 power setting without water, attached to a P5 NEWTRON scaler (ACTEON). The orifice was enlarged using micro-openers (DENTSPLY Maillefer). The previous obturation was removed using retreatment rotary files (Endostar, Poldent). Coronal pre-flaring was done using 25/.08 TF files (KaVo Kerr). A glide path and patency were established in all the canals with a 10/.02 K-flex file (KaVo Kerr) using a watch winding motion and with an electronic apex locator (Apex ID, KaVo Kerr) attached to simultaneously record the EWL (electronic working length; Table I). The glide path was then widened using an intermediate size file of 12/.02 (Endostar) and a 15/.02 K-flex file (KaVo Kerr) in balanced force motion. The initial master apical file was then selected for each canal, sequentially placing larger K-flex files and checking the file that bound within 3–4 mm short of the EWL. For the mesiobuccal canal, it was 30/.02; for the mesiolingualcanal, it was 35/.02; and for the distal canal, it was 40/.02. The canals were then sequentially enlarged with K-flex files in the M4 Safety handpiece (KaVo Kerr) at 5,000–6,000 rpm.
After every 0.5–1.0 mm of advancement, the canal was irrigated with a mixture of 6% sodium hypochlorite (CanalPro, COLTENE) and HEBP (1-hydroxyethylidene-1,1-bisphosphonate) or etidronic acid Twin Kleen (Maarc Dental) at a dilution ratio of 0.9 g (two capsules) of HEBP in 10 ml of sodium hypochlorite. The solution follows the concept of continuous chelation that either prevents or minimises the formation of the smear layer, and it is especially effective in the critical apical region. The solution was placed into the canal with a 30-gauge double-side-vented needle (Master Clean 2, Ident) at a flow rate of 1 ml per fifteen seconds (Table II). Then negative pressure irrigation was performed with 10 ml per canal in two phases using EndoVac (KaVo Kerr).
Patency was checked with a 10/.02 K-flex file with the electronic apex locator attached to record any change in the working length that may have resulted from preparation of the canals. The distal canal was ovoid in shape; hence, the canal body shaping was done using 15/.02 H-type files in the circumferential filing technique. After the preparation of the canals to the initial master apical file size, the canals were sequentially merged with 4% taper rotary files. I used Endostar Azure rotary files (Poldent) and shaped to the respective final master apical file (Table I). Since the oozing of blood had not stopped and the canals were not dry after the cleaning and shaping of the canals, it was decided to place calcium hydroxide into the canals and cover the perforation with PTFE tape that had been soaked in 2% chlorhexidine for ten minutes, and the access was sealed with a light-polymerising temporary composite.
The patient was recalled after ten days. At the second appointment, the temporary restoration and the tape were removed. The pulp chamber and the canals were irrigated with saline to wash off the excess calcium hydroxide. The canals and pulp chamber were treated with 17% EDTA for one minute, followed by ultrasonic activation for thirty seconds with an Irrisafe tip (ACTEON) attached to a P5 NEWTRON scaler unit. Then the canals were irrigated according to the continuous chelation concept using a mixture of HEBP and sodium hypochlorite in negative pressure irrigation using EndoVac (10 ml per canal) in two phases. Figure 4 shows the pulp chamber just after the irrigation protocol. The canals were then dried using 70% isopropyl alcohol and paper points.
AH Plus sealer (Dentsply Sirona) was applied to the canal walls using the respective gutta-percha master cones. The gutta-percha cones were pre-sterilised by placing them in 2% chlorhexidine for ten minutes. The canals were then obturated by Schilder’s warm vertical compaction using Touch ’n Heat (SybronEndo), heat carriers and Buchanan hand pluggers (SybronEndo). After the corono-apical downpacking, the rest of the canal was back-filled with thermoplastised gutta-percha using a System B gun (SybronEndo). The backfilling was done keeping it 3 mm short of the orifice to allow the core to form orifice plugs that would increase the retention and coronal seal by increasing the surface area for bonding (Fig. 5). Then the pulp chamber was cleaned with 70% isopropyl alcohol and treated with 17% EDTA for one minute.
Since the defect was large, it was decided to place a PRF membrane beneath the MTA. Blood (10 ml) was drawn from the cubital region and collected in two Vacutainer tubes without anti-coagulant. The tubes were then immediately placed in a portable centrifuge. The centrifuge was set at 3,000 rpm for ten minutes. The canal orifices were protected from accidental blocking with MTA by packing sterile PTFE in them. The middle layer of PRF (the bottom layer contains red blood cells and the top layer contains platelet-poor plasma) was isolated with a pair of sterile tweezers and placed in a sterile membrane-forming box then carried to the perforation site and adapted lightly. Then MTA Plus (Prevest Denpro) was mixed and placed in increments and adapted all along the margins and into the defect. Finally, a moist sterile gauze was placed over the MTA, and the access was closed with a light polymerising temporary composite for 24 hours.
At the next appointment, the temporary restoration was removed, and the hardness of the MTA was checked. The margins of the defect were also closely checked under the dental operating microscope to rule out any leakage. Finally, a thin layer of light-polymerising glass ionomer cement liner was placed over the MTA. Etching and bonding followed, and the core was placed using a bulk fill composite in a SonicFill handpiece (KaVo Kerr, Figs. 6a–g).
The patient was evaluated at a six- and 14-month interval. Clinically, the probing depth was fine and there was no loss of attachment but firm attached gingiva. Radiographically, both the six-month and 14-month radiographs (Figs. 7a & b) showed improvement in relation to the periapical and furcation radiolucency when compared with the pretreatment intra-oral periapical radiograph.
Discussion
The success of an endodontic treatment is estimated to be high, over 98% for initial treatment and over 95% in retreatment cases, according to peer-reviewed literature and evidence-based studies, but failures do happen for a spectrum of reasons, which include biological and technical aspects that ultimately lead to an increased bacterial load within the canal space. An endodontic treatment may be deemed to have failed if the treated tooth presents with symptoms or periapical radiolucency or both. The various reasons for failure could be coronal leakage from a poor or inadequate post-endodontic restoration, a missed canal or anatomy, incomplete instrumentation of the canals and spaces, insufficient irrigation and disinfection of the canals.
To achieve adequate cleaning in the apical region, adequate apical sizes should be created without sacrificing the precious radicular dentine. In this regard, the sequential flaring and enlargement of the canal (hybrid flaring technique) at different levels has a substantial advantage in having the benefits of all the techniques and combining them to enhance the outcome of the case. The stages of the instrumentation protocol are as follows:
- The canals are located using 8/.02 and 10/.02 K-type files in a watch-winding motion. The files are not advanced any further than the beginning of the first curvature.
- Coronal pre-flaring is performed with 6–8% taper orifice shapers in rotary motion until the beginning of the curvature of the canal. The canals are flooded with irrigation solution (sodium hypochlorite and HEBP).
- The glide path and patency are established with 8 and 10 K-type files in a watch winding motion with an electronic apex locator attached to record the EWL.
- Widening of the glide path is done using 12/.02 and 15/.02 K-type files in manual balanced force motion, and a 10/.02 K-type file is used in between to confirm patency and irrigation solution is exchanged between files.
- The initial master apical file is selected from any 2% taper K-type file larger than 15 that binds 3–4 mm short of the EWL.
- Apical enlargement is done using sequential 2% taper K-type file larger than 15 to shape the canal either manually using the balanced force technique or attached to an engine-driven handpiece that uses alternating clockwise and anticlockwise motion until the initial master apical file size is reached. In between, after 0.5–1.0 mm of advancement with each file, patency is checked with a 10/.02 K-type file and the irrigation solution is exchanged. Here either the traditional ISO sizes may be followed in the sequence or intermediate files may be used to reduce the percentage jump in the file size (the latter is recommended), for example 12/.02, 15/.02, 17.5/.02, 20/.02, 22.5/.02, 25/.02, 27.5/.02, 30/.02. This allows for a smoother transition to the next size, aiding effortless shaping of the canal along with maintenance of the original path and anatomy of the canal.
- The apical preparation is shaped and flared with 4% taper files until the initial master apical file size is reached. The final shaping file becomes the final master apical file (same tip size but a taper of 4%).
The hybrid flaring technique of instrumentation has the following benefits:
- It addresses the critical apical third adequately, both mechanically and biologically.
- It helps achieve adequate apical size, reducing the risk of iatrogenic errors such as ledge formation, straightening of the canal, file separation and transportation, common with tapered rotary files used in continuous rotary motion, since only 2% of files are used in balanced force motion, where the primary cutting happens in the anticlockwise turn.
- It promotes less packing of debris in the apical region, as coronal pre-flaring precedes apical preparation and most of the preparation is with either hand files or files attached to an engine-driven handpiece. Coronal pre-flaring actually helps reduce the active length of the subsequent file meant to shape the apical region by flaring the canal until the beginning of the first curvature and prevents instrument failure by torsion.
- The final preparation with 4% taper files is made less stressful to the canal walls because of less engagement of dentine and a wider glide path, and adds to rotary safety.
- The final taper of 4% adequately aids in placement of the irrigation needle tip to within 1 mm of the EWL, activation of the irrigant with sonic and ultrasonic aids, and placement of the irrigation needle tip to within 1 mm of the EWL for negative pressure irrigation.
Magnification and ergonomic use of the dental operating microscope are vital in such a clinical scenario. Working at high magnification, 16× and above, needs outstanding eye–hand–mirror coordination, and precision is paramount in dealing with a similar clinical scenario.
Many materials to seal iatrogenic perforations have been tested. The ideal material used to repair perforations should be non-toxic, non-absorbable and radiopaque, should possess antimicrobial properties and should seal against microleakage through the margins of the perforation. MTA has all of these characteristics and has been employed extensively in regenerative endodontics. It is the material of choice for managing perforation repair and for regenerative endodontics. This can be attributed to its biocompatibility, low induction of inflammation, low solubility, and capacity to create a seal between the pulp chamber and periodontal tissue. The most important attributes are its antimicrobial properties and high pH of 12.5, which promote growth of the cementum and formation of bone, in turn allowing regeneration of the periodontal ligament around the site of injury. When combined with PRF in large defects, both may act in synergy in the regenerative process. PRF contains platelets, growth factors and cytokines that might enhance the healing potential of both soft and hard tissue. Though there is a lack of literature support regarding the synergistic use of MTA and PRF, owing to the osteoconductive and inductive nature of MTA and the autologous nature of PRF, their combination seems promising.
Follow-up
Though the prognosis of the case initially appeared to be poor, the six- and 14-month follow-ups showed a favourable outcome. The periapical healing was considerable and the furcation defect too had responded well. It appears that adequate cleaning and shaping of the canals and the use of a detailed irrigation and disinfection protocol and a biocompatible, autologous and tissue-conductive material to seal the perforation acted in synergy to address the biological aspect of the treatment. The aspect that remained to be evaluated was the functional one, for which there was need for more extensive follow-up.
Conclusion
From the case described, it may be concluded that, since it is in evidence that the initial endodontic treatment has a higher percentage of success compared with retreatment, all efforts should be made to deliver the best care on the first go. It was also learnt that whenever possible a decision should be taken to refer the case to a specialist rather than attempt treatment with a limited knowledge and armamentarium, as the Hippocratic oath says, “at least to do no harm”. Lastly, sometimes the worst appearing case may be salvageable, and adequate cleaning, shaping, irrigation and disinfection, the use of biocompatible regenerative materials and above all the host response play a vital role.
Editorial note: A list of references is available from the publisher. This article was published in roots—international magazine of endodontics Vol. 16, Issue 2/2020.
Tags:



 Austria / Österreich
Austria / Österreich
 Bosnia and Herzegovina / Босна и Херцеговина
Bosnia and Herzegovina / Босна и Херцеговина
 Bulgaria / България
Bulgaria / България
 Croatia / Hrvatska
Croatia / Hrvatska
 Czech Republic & Slovakia / Česká republika & Slovensko
Czech Republic & Slovakia / Česká republika & Slovensko
 France / France
France / France
 Germany / Deutschland
Germany / Deutschland
 Greece / ΕΛΛΑΔΑ
Greece / ΕΛΛΑΔΑ
 Hungary / Hungary
Hungary / Hungary
 Italy / Italia
Italy / Italia
 Netherlands / Nederland
Netherlands / Nederland
 Nordic / Nordic
Nordic / Nordic
 Poland / Polska
Poland / Polska
 Portugal / Portugal
Portugal / Portugal
 Romania & Moldova / România & Moldova
Romania & Moldova / România & Moldova
 Slovenia / Slovenija
Slovenia / Slovenija
 Serbia & Montenegro / Србија и Црна Гора
Serbia & Montenegro / Србија и Црна Гора
 Spain / España
Spain / España
 Switzerland / Schweiz
Switzerland / Schweiz
 Turkey / Türkiye
Turkey / Türkiye
 UK & Ireland / UK & Ireland
UK & Ireland / UK & Ireland
 International / International
International / International
 Brazil / Brasil
Brazil / Brasil
 Canada / Canada
Canada / Canada
 Latin America / Latinoamérica
Latin America / Latinoamérica
 USA / USA
USA / USA
 China / 中国
China / 中国
 Pakistan / Pākistān
Pakistan / Pākistān
 Vietnam / Việt Nam
Vietnam / Việt Nam
 ASEAN / ASEAN
ASEAN / ASEAN
 Israel / מְדִינַת יִשְׂרָאֵל
Israel / מְדִינַת יִשְׂרָאֵל
 Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
 Middle East / Middle East
Middle East / Middle East








































Appreciate your suggestion. The term “iatrogenic” has been widely used. Though more specific terms as “odontoiatrogenic” and “dentistogenic” should be accepted by the fraternity. Also if continued on the same line there should be some different term for mishaps of “surgeons origin”, but they too widely use the same term in similar clinical situations.
With all due respect, the term “iatrogenic” (Greek), means of physician origin; more appropriately would be the use of the term “odontotiatrogenic”, which means of dentist origin, as I do not believe there are any physicians perforating the floor of teeth these days.